 However, the filling pattern is interrupted by two elements, chromium (Cr) and copper (Cu), both of which have partially filled 4s-subshells. On the other hand, titanium has 22 electrons and two electrons in its 3d-subshell, resulting in an electron configuration of 3d2 4s2. For example, scandium (Sc) has 21 electrons and just one electron in its 3d-subshell, resulting in an electron configuration of 3d1 4s2. The next ten elements in the period belong to the d-block, and their electrons are added to the inner 3d-subshell one by one as we move across the period. As a result, the 3d-subshells fill up after the 4s-subshells, which is another tricky exception to the filling pattern that you need to remember. However, 3d-subshells are an exception to this rule since they have slightly higher energy levels than 4s-subshells. Subshells typically fill up in a specific order, from the lowest to the highest energy level, which typically follows the pattern of lowest to highest numbers. The valence electrons of these elements are found in the 4s-subshell, and their 3d-subshells are empty. The first two elements in period 4, potassium (K) and calcium (Ca), are located in the s-block of the periodic table. To illustrate this, let's take a closer look at the first row of transition metals (period 4) and highlight it below. These electrons fill up the d-subshell gradually, although there are some exceptions. As we move across the period in the periodic table, each transition metal has one more electron than the previous one. For instance, the highest energy subshell for p-block elements is a p-subshell. An element's position in the periodic table indicates the highest energy subshell where its electrons are found. It's important to remember that electrons are organized into shells and subshells, with four different types: s-, p-, d-, and f-subshells. Electron configuration of transition metal atomsĪll transition metals can be found in the d-block of the periodic table, meaning that their valence electrons are located in a d-subshell. We'd recommend checking them out first to learn the basics of electron shells, sub-shells, orbitals, and filling rules. This section probably won't make much sense if you haven't read Electron Shells and Electron Configuration. This will also help explain why certain members of the d-block aren't classified as transition metals. We'll start with their electron configuration as atoms, and then look at how this changes as they form ions. Electron configuration of transition metals The transition metals can be found in groups 3-12 and periods 4-7, although what's more crucial is that they can be easily located in the periodic table. The highest energy subshell in the d-block is always a d-subshell. You can find the transition metals in the middle of the d-block section of the periodic table, which acts as a link between the s- and p-blocks. However, for this article, we will be focusing solely on the elements highlighted in blue above, as defined by IUPAC. Additionally, there are two other groups of elements called the lanthanides and actinides, with atomic numbers 57-71 and 89-103 respectively, which are sometimes referred to as inner transition metals. 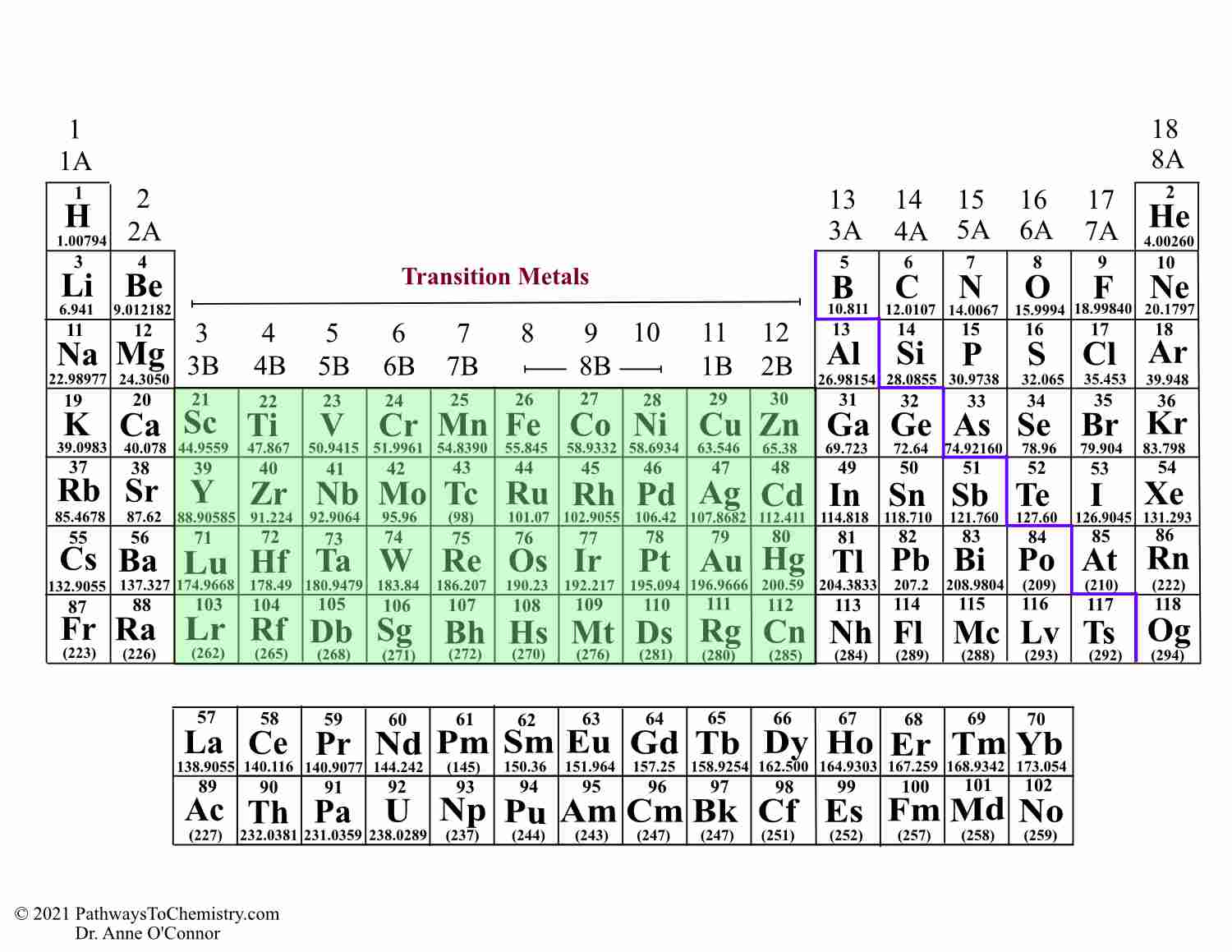
The reason for this will be discussed later on. It's important to note that not all elements in the d-block are transition metals, despite the term being used interchangeably. Periodic table of elements with transition metals highlighted 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
